Unlocking mRNA Vaccine Secrets: How They Evade Cellular Defenses
Table of Contents
- Unlocking mRNA Vaccine Secrets: How They Evade Cellular Defenses
- mRNA Therapies: A New Frontier in Genetic Medicine
- Cellular Gatekeepers: Identifying Key Players in mRNA processing
- The Nobel-Winning Secret: How mRNA Vaccines Bypass Immune Detection
- Proton Power: The Unexpected Role of Ions in Immune Signaling
- Implications for Future therapies
- The Path Forward: Early Intervention and Targeted Treatments
archynetys.com – In-depth analysis of groundbreaking research in mRNA technology.
mRNA Therapies: A New Frontier in Genetic Medicine
Messenger RNA (mRNA) technology has revolutionized medicine, particularly with the rapid advancement and deployment of mRNA vaccines against COVID-19. However, the intricate mechanisms by which these vaccines navigate and interact with our cells have remained somewhat mysterious. Now, a groundbreaking study sheds light on these processes, revealing key factors that influence mRNA delivery, stability, and degradation within cells. This finding not only deepens our understanding of existing mRNA vaccines but also paves the way for innovative genetic therapies and personalized treatments.
Cellular Gatekeepers: Identifying Key Players in mRNA processing
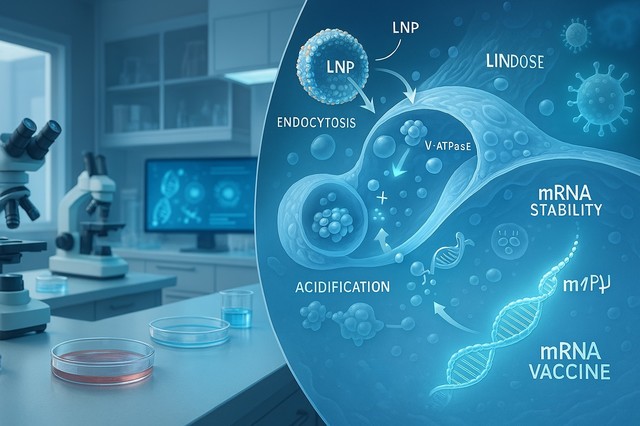
Researchers have identified three crucial cellular components that govern the fate of mRNA within the body. These findings offer a novel model for understanding how mRNA therapies function and how their effectiveness can be enhanced.
- Heparan Sulfate Proteoglycans (HSPGs): These cell surface molecules act as initial attractors, drawing lipid nanoparticles (LNPs), the delivery vehicles for mRNA, to the cell. HSPGs essentially facilitate the first step of mRNA entry.
- Vacuolar (V)-ATPases: Located within endosomes, these proton pumps create an acidic surroundings that destabilizes the LNP membrane, enabling the release of mRNA into the cytoplasm. This step is critical for the mRNA to access the cellular machinery needed for protein synthesis.
- TRIM25 Protein: This protein functions as a cellular defense mechanism, binding to and degrading foreign mRNA, thus preventing its action. TRIM25 represents a significant hurdle that mRNA vaccines must overcome.
The Nobel-Winning Secret: How mRNA Vaccines Bypass Immune Detection
A pivotal aspect of the study reveals how mRNA vaccines, particularly those utilizing a modified nucleoside called N1-methylpseudouridine (m1Ψ), manage to evade detection by TRIM25. This modification, recognized with the Nobel Prize in Medicine in 2023, prevents TRIM25 from binding to the mRNA, thereby increasing its stability and translational efficiency.This discovery elucidates the critical importance of this modification for the success of mRNA therapies.
The incorporation of m1Ψ into mRNA is a game-changer. it allows the mRNA to persist longer within the cell, leading to increased protein production and a stronger immune response.
Proton Power: The Unexpected Role of Ions in Immune Signaling
The research also highlights the surprising role of proton ions in triggering cellular defense responses. When LNPs disrupt the endosomal membrane, protons are released into the cytoplasm, activating TRIM25. These ions act as an alarm signal, alerting the cell to the presence of foreign RNA and initiating protective measures. This is the first study to demonstrate that proton ions serve as immune signal molecules, providing new insights into how cells defend themselves against exogenous RNA.
Implications for Future therapies
These findings have profound implications for the future of mRNA therapies. By understanding how cells react to vaccines and how to effectively navigate cellular defense mechanisms,researchers can develop more potent and targeted treatments for a wide range of diseases. For example, optimizing LNP design to control proton release could enhance mRNA delivery and reduce unwanted immune responses. Furthermore, strategies to inhibit TRIM25 activity could further improve the efficacy of mRNA vaccines and therapies.
“In order to improve the mRNA therapies, we need to understand how the cells react to vaccines. It is essential to find ways to avoid cell defense mechanisms and to use the endomal system effectively,” Dr. Kim V. Narry, director of the Centre for RNA research within the Institute for Fundamental Sciences (IBS).
The Path Forward: Early Intervention and Targeted Treatments
This research not only paves the way for more effective mRNA vaccine delivery but also provides a foundation for developing future RNA therapies. The results underscore the importance of early intervention and offer new directions for more effective treatments in various diseases. As mRNA technology continues to evolve, these insights will be crucial for unlocking its full potential and revolutionizing healthcare.