Novel Drug Combination Shows Promise in Reducing Liver Fat
Table of Contents
A recent study indicates that a combination therapy significantly reduces liver fat content in patients with presumed metabolic dysfunction-associated steatohepatitis (MASH).
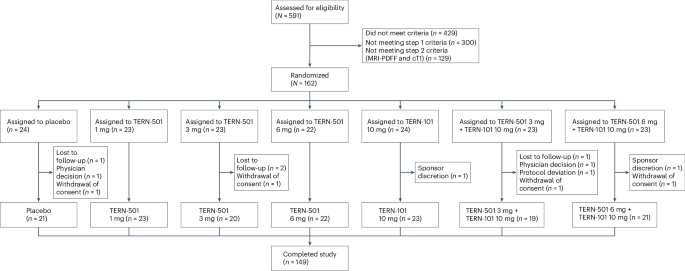
A phase 2a clinical trial, known as the DUET study, has revealed promising results for a new treatment approach targeting metabolic dysfunction-associated steatohepatitis (MASH). The 12-week study, published in Nature Medicine, investigated the efficacy of TERN-501, a thyroid hormone receptor β agonist, both as a standalone treatment and in conjunction with TERN-101, a farnesoid X receptor agonist.
The findings demonstrated that both TERN-501 monotherapy and the TERN-501/TERN-101 combination led to dose-dependent reductions in liver fat content compared to a placebo. the study focused on patients with presumed MASH, a condition characterized by liver inflammation and damage due to fat buildup.
Key Findings of the DUET Study
“Treatment with TERN-501… resulted in dose-dependent reductions in liver fat content as compared to placebo.”
The DUET study provides encouraging evidence for the potential of TERN-501, particularly when combined with TERN-101, as a therapeutic intervention for MASH. Further research is needed to confirm these findings and assess the long-term safety and efficacy of this treatment approach.
Implications for MASH Treatment
MASH, formerly known as nonalcoholic steatohepatitis (NASH), is a progressive liver disease affecting a significant portion of the global population. Currently, there are limited treatment options available, highlighting the urgent need for novel therapies. The results of the DUET study offer hope for the development of more effective treatments for MASH and related metabolic disorders.
Sources
- Nature Medicine: Two: 10.1038/S41591-025-03722-7
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK): https://www.niddk.nih.gov/health-information/liver-disease/nafld-nash
- World Health Organization (WHO): https://www.who.int/news-room/fact-sheets/detail/hepatitis-b
- Centers for Disease Control and prevention (CDC): https://www.cdc.gov/nchs/products/databriefs/index.htm
- European Association for the Study of the Liver (EASL): https://www.easl.eu/nafld/