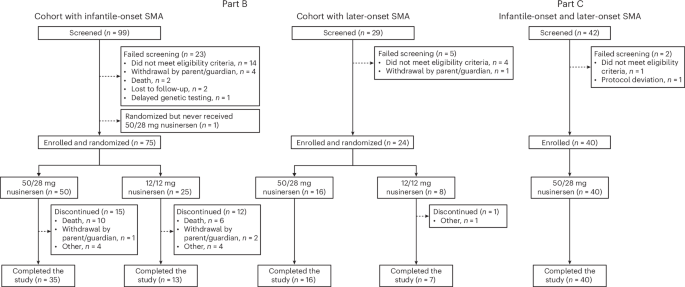
Participant disposition
Table of Contents
- Participant disposition
- Primary outcomes
- Secondary outcomes
- Part B cohort with infantile-onset SMA: motor function
- Part B cohort with infantile-onset SMA: neurodegeneration
- Part B cohort with infantile-onset SMA: event-free survival
- Part B cohort with infantile-onset SMA: other key outcomes
- Part B cohort with later-onset SMA (treatment-naive participants, n = 24 randomized 2:1): motor function
- Part B cohort with later-onset SMA: neurodegeneration
- Part C previously-treated participants (n = 40 open-label): motor function
- Part C previously-treated participants: neurodegeneration
- Parts B and C: pharmacokinetics
- Safety
- Exploratory outcomes and post hoc analyses
Parts B and C of the DEVOTE trial screened 170 patients and enrolled 139 participants between 12 November 2020 and 8 August 2023. In the Part B cohort with infantile-onset SMA, 50 participants received 50/28 mg nusinersen (35 (70%) completed the trial) and 25 received 12/12 mg (13 (52%) completed). The matched sham arm from ENDEAR included 20 participants. For the cohort with later-onset SMA, 16 received 50/28 mg (all completed the trial) and eight received 12/12 mg (seven (87.5%) completed). Comparator groups for the DEVOTE cohort with later-onset SMA included matched participants from CHERISH who had received sham (16 participants) and 12/12 mg nusinersen (32 participants). In Part C, 40 participants were enrolled (all completed the trial). Screening failures and treatment discontinuations are shown in Fig. 1.
The Part B cohort with infantile-onset SMA was compared with a matched group of 20 participants from ENDEAR who had received sham. The Part B cohort with later-onset SMA was compared with two matched groups of participants from CHERISH who had received sham (n = 16) and 12/12 mg nusinersen (n = 32).
Participants’ characteristics across all DEVOTE cohorts and matched comparator groups are shown in Table 1.
Participants in the cohort with infantile-onset SMA in Part B had a mean age at first dose of 18.4 (s.d. = 9.15) weeks and disease duration of 9.6 (s.d. = 5.26) weeks in the 50/28 mg group compared with 22.0 (s.d. = 7.96) weeks and 11.1 (s.d. = 4.92) weeks, respectively, in the matched sham group. All had two SMN2 copies. Prespecified matching helped minimize imbalance between groups; however, DEVOTE participants were younger at symptom onset with lower Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders (CHOP-INTEND) scores indicating more severe disease relative to the ENDEAR matched sham group.
In the cohort with later-onset SMA in Part B, nearly all participants (22/24 (92%)) had three SMN2 copies and a median age of 6.1 (Q1, 4.2; Q3, 8.2) years at first dose in the 50/28 mg group compared with 5.7 (Q1, 2.8; Q3, 8.5) in the 12/12 mg group. The demographic characteristics and disease characteristics of the DEVOTE treatment groups and CHERISH comparator groups were generally similar. However, baseline Hammersmith Functional Motor Scale Expanded (HFMSE) scores, Revised Upper Limb Module (RULM) scores and World Health Organization (WHO) motor milestones were lower in the DEVOTE 12/12 mg group compared with the 50/28 mg, CHERISH matched sham and CHERISH matched 12/12 mg groups.
Of the 40 participants enrolled in Part C, two participants (5%) had infantile-onset SMA (both had three SMN2 copies) and 38 (95%) had later-onset SMA. Part C participants had 1–4 SMN2 copies, were aged 4–65 years at the time of first study dose and had received 12/12 mg for a median of 3.9 years before transitioning to 50/28 mg in DEVOTE.
Primary outcomes
For the primary endpoint, assessed in the Part B cohort with infantile-onset SMA, the 50/28 mg group experienced a statistically significant improvement in CHOP-INTEND at day 183 compared with the ENDEAR matched sham group, accounting for death using the joint-rank test (difference in ranks was 26.06 (95% confidence interval (CI) = 17.9 to 34.2); P < 0.0001). The least-squares mean (LSM) change was a 15.1-point (95% CI = 12.4 to 17.8) improvement with 50/28 mg and an 11.1-point (95% CI = −15.9 to −6.2) worsening in the matched sham (difference, 26.19 (95% CI = 20.7 to 31.74)) (Fig. 2a and Table 2).
aThe primary analysis was performed using ranking based on the CHOP-INTEND score at day 183 or day of death and fitting an ANCOVA model adjusting for disease duration and baseline CHOP-INTEND to determine the difference in ranks via the joint-rank test. MI was used to impute missing data. bResults are from an ANCOVA model with adjustment for participants’ disease duration, baseline log plasma NfL and baseline CHOP-INTEND score. MI was used to impute missing data. In the table, the LSGM ratio from the ANCOVA is shown; the P value was determined using the joint-rank test. cThe P value was determined using the log-rank test stratified according to disease duration (≤12 weeks or >12 weeks). P values were obtained by comparing the two treatment groups indicated per row in each table above each chart. Comparisons between 50/28 mg and 12/12 mg, and between 50/28 mg and matched sham, were performed as separate analyses. In addition to the P value, rankings, changes from baseline in actual scores and ratios are shown. For the EFS and OS, comparisons were similarly made between the two treatment groups. Because of hierarchical testing, none of the time-to-event endpoints were considered statistically significant. Where P < 0.05 but hierarchical testing had stopped, the P value was considered nominally significant and is denoted. aP value was considered nominally significant. Study day 1 was the baseline. All statistical tests were two-sided. LSGM, least-squares geometric mean.
Secondary outcomes
Part B cohort with infantile-onset SMA: motor function
The proportion of Hammersmith Infant Neurological Exam Section 2 (HINE-2) responders at day 183 was significantly higher in the 50/28 mg group compared with the ENDEAR matched sham group (P < 0.0001), as was the magnitude of improvement in mean HINE-2 score at day 183 (P < 0.0001) (Table 2 and Extended Data Fig. 1).
While the study was not powered to detect statistically significant differences between the active treatment groups, secondary endpoints were included in the hierarchy of testing. For CHOP-INTEND, the LSM ranked score from baseline at day 302 was numerically higher in the 50/28 mg group than the 12/12 mg group, indicating greater improvement on CHOP-INTEND when accounting for death; the LSM actual score was numerically greater in the 12/12 mg group (Table 2 and Extended Data Fig. 2). For HINE-2, both the ranked and actual scores trended in favor of the 50/28 mg group.
Part B cohort with infantile-onset SMA: neurodegeneration
Reduction in plasma neurofilament light chain (NfL) levels from baseline at day 183 were significantly greater in the 50/28 mg group (94% reduction) compared with the ENDEAR matched sham group (30% reduction) (P < 0.0001), indicating a substantial slowing of neurodegeneration in response to high-dose nusinersen (Table 2 and Fig. 2b). More rapid reductions in NfL levels were observed in the 50/28 mg group (88%) compared with the 12/12 mg group (77%) at day 64 (nominally significant P = 0.0050; joint-rank test) (Fig. 2b).
Part B cohort with infantile-onset SMA: event-free survival
The 50/28 mg group experienced a lower risk of death or permanent ventilation relative to the ENDEAR matched sham group (hazard ratio (HR) = 0.322; nominal P = 0.0006; log-rank test); results were similar for survival (Table 2 and Fig. 2c). The 50/28 mg group experienced a trend toward a lower risk of death or permanent ventilation relative to the 12/12 mg group (HR = 0.701; P = 0.2775; log-rank test); results were again similar for survival.
Part B cohort with infantile-onset SMA: other key outcomes
The proportion of participants hospitalized and time in hospital attributed to serious adverse events (SAEs), the proportion of participants experiencing serious respiratory events and the proportion of time participants used ventilation support were all numerically lower in the 50/28 mg group compared with the 12/12 mg group (Extended Data Table 1). Of those participants with an opportunity to improve (those with a suck or swallow deficit on HINE-1 at baseline (n = 23 in the 50/28 mg group and n = 11 in the 12/12 mg group), 39% of the 50/28 mg group and 9% of the 12/12 mg group experienced improvement from baseline to day 302. Additionally, a numerically larger proportion of investigators and caregivers of participants in the 50/28 mg group reported a response of ‘much improved’ or ‘very much improved’ from baseline to day 302 on the Clinical Global Impression of Change than was reported for the 12/12 mg group.
Part B cohort with later-onset SMA (treatment-naive participants, n = 24 randomized 2:1): motor function
Participants in the 50/28 mg group experienced numerically greater improvements on both HFMSE and RULM from baseline to day 302 compared with the 12/12 mg group (Fig. 3a,c). However, these trends were variable over time, which is consistent with the limited sample size. More consistent differentiation was observed between the 50/28 mg group and prespecified matched comparator groups from CHERISH (sham and 12/12 mg), both of which provided larger sample sizes (Figs. 3b,d).
Data in the tables represent the mean (s.d.). a,cStudy day 1 was the baseline and MI was used for missing data. b,dData were based on MI and ANCOVA with treatment as a fixed effect and adjustment for each participant’s age at first dose, baseline log plasma NfL and baseline HFMSE or RULM score. CHERISH matched 12/12 mg and sham groups indicate prespecified subsets of participants matched to the characteristics of the DEVOTE 50/28 mg group. Analysis days 183 and 279 in DEVOTE correspond with days 169 and 274 in CHERISH, respectively.
Part B cohort with later-onset SMA: neurodegeneration
Greater reductions in plasma NfL levels were observed in the 50/28 mg group compared with the 12/12 mg group at day 64 (nominally significant P = 0.0495) (Extended Data Fig. 4).
Part C previously-treated participants (n = 40 open-label): motor function
After transition to the 50/28 mg regimen, participants experienced a mean improvement on HFMSE of 1.8 points (s.d. = 3.99) from baseline to day 302, with a 2.3-point (s.d. = 3.95) mean improvement in the adult subgroup (n = 24) (Supplementary Fig. 1a). Overall, 53% of participants (n = 20/38) experienced an increase in HFMSE score from baseline to day 302 (Extended Data Fig. 5a).
On RULM, participants experienced a mean improvement of 1.2 points (s.d. = 2.14) from baseline to day 302, with a 0.9-point (s.d. = 1.89) improvement in the adult subgroup (Supplementary Fig. 1b). Of those with an opportunity to improve (a score below the maximum possible at baseline; n = 26), 16 (62%) experienced an increase in RULM score from baseline to day 302 (Extended Data Fig. 5b).
Part C previously-treated participants: neurodegeneration
Given prior treatment with nusinersen, older age and more advanced disease, plasma NfL levels were low at baseline in DEVOTE Part C (5.2 pg ml−1; Table 1) and remained low throughout the study.
Parts B and C: pharmacokinetics
The 50/28 mg regimen in both Part B cohorts with infantile-onset and later-onset SMA achieved higher plasma and cerebrospinal fluid (CSF) trough concentrations than the 12/12 mg regimen (Supplementary Notes (Results, p. 24) and Supplementary Figs. 2 and 3). Day 15 CSF trough concentrations of nusinersen were approximately twofold higher after the first dose of 50 mg compared with the first dose of 12 mg nusinersen (Supplementary Fig. 4). Although the increase in CSF exposures was less than dose-proportional, 50/28 mg achieved higher CSF trough levels more quickly (one dose of 50 mg versus three doses of 12 mg) (Supplementary Fig. 5). Plasma concentrations of nusinersen increased approximately linearly with dose across all measured time points. In Part C, pre-dose CSF concentrations of nusinersen increased rapidly after the 50-mg loading dose on day 1; the higher concentrations were maintained during the maintenance dose period (Supplementary Notes: Results, p. 25). No nusinersen accumulation within plasma was observed after multiple doses in Parts B and C.
Safety
The safety profile of the 50/28 mg regimen was broadly consistent with the known safety profile of the 12/12 mg regimen.
Across Part B, 66 treatment-naive participants (50 infantile-onset, 16 later-onset) received 50/28 mg nusinersen, for a total of 50.06 participant-years; 33 treatment-naive participants (25 infantile-onset, eight later-onset) received 12/12 mg nusinersen for 21.44 participant-years. In Part C, 40 participants received 50/28 mg nusinersen for a total of 34.52 participant-years.
Across all groups treated with 50/28 mg in Parts B and C, most adverse events (AEs) were mild to moderate in severity and did not lead to treatment discontinuation; all AEs that led to study withdrawal were fatal AEs and were assessed as unrelated to treatment by the investigator (Table 3). Many of the commonly reported AEs were consistent with events occurring in the natural history of SMA, common conditions in the general population or events observed in the context of a lumbar puncture procedure. Few AEs were considered related to study treatment across all groups (<7% across all Part B; 20% in both Part C and the ENDEAR matched sham). Across all groups, only one participant in the Part B infantile-onset 12/12 mg group experienced a treatment-related SAE (respiratory failure).
In the Part B cohort with infantile-onset SMA of 50 participants, the most common AEs (≥15% of participants) in the 50/28 mg group were pneumonia (10 (20%)), respiratory failure (10 (20%)), pyrexia (9 (18%)), coronavirus disease 2019 (COVID-19) (8 (16%)) and upper respiratory tract infection (8 (16%)). In the ENDEAR matched sham group, there was a higher incidence of respiratory failure (8 (40%)), pyrexia (9 (45%)) and upper respiratory infection (5 (25%)), and a lower incidence of pneumonia (1 (5%)) and COVID-19 (0 (0%)) compared with the Part B infantile-onset 50/28 mg group. COVID-19 was not relevant at the time of ENDEAR; the higher incidence of pneumonia is discussed below. Overall, the frequency of participants reporting severe AEs in the 50/28 mg (22/50 (44%)) and 12/12 mg groups (14/25 (56%)) was lower compared with the ENDEAR matched sham group (18/20 (90%)). The frequency of participants reporting SAEs was lower in the 50/28 mg group (30/50 (60%)) compared with the 12/12 mg (18/25 (72%)) and ENDEAR matched sham groups (19/20 (95%)) (Table 3). There were no treatment-related SAEs in the 50/28 mg group. SAEs that occurred in ≥10% participants in the 50/28 mg group, which also had a frequency ≥5% in the 50/28 mg group compared with the ENDEAR matched sham, were pneumonia (seven (14%) versus 1 (5%)) and pneumonia aspiration (seven (14%) versus one (5%)). Of seven participants with pneumonia aspiration in the 50/28 mg group, five experienced the event before day 35, with two events occurring on day 1. All events were assessed as unrelated to study treatment by the investigator.
The frequency of fatal AEs was lower in participants treated with nusinersen across all groups compared with matched sham (Table 3). All fatal AEs occurred in the Part B cohort with infantile-onset SMA, and the frequency in the 50/28 mg group (10/50 (20%)) was lower than in the 12/12 mg (6/25 (24%)) or ENDEAR matched sham groups (11/20 (55%)).
Safety profiles in the Part B later-onset and Part C cohorts were similar to that in the Part B cohort with infantile-onset SMA. Across Parts B and C, there were no AEs of meningitis, hydrocephalus, thrombocytopenia or renal/liver failure reported, and no clinically relevant trends related to nusinersen in laboratory evaluations, electrocardiograms or vital signs.
Exploratory outcomes and post hoc analyses
Exploratory outcomes and post hoc analyses are described in the Supplementary Notes: Results, p. 26.