The Physics Paradox: When Chaos Becomes Clarity
The experiment began when MIT researchers gradually increased the power of a laser traveling through a multimode optical fiber, a setup known for scattering light into unpredictable patterns. At higher intensities, the light was expected to grow more disordered—until it didn’t. Instead, as the power neared the fiber’s damage threshold, the scattered rays coalesced into a single, focused beam capable of high-speed imaging through living tissue.
Officials involved in the study noted that the prevailing assumption in the field was that increasing power in this type of laser would lead to greater chaos. However, the team found that under specific conditions, the light could self-organize into a stable, high-resolution beam. This effect required the laser to enter the fiber at a zero-degree angle while operating at the edge of the fiber’s capacity.
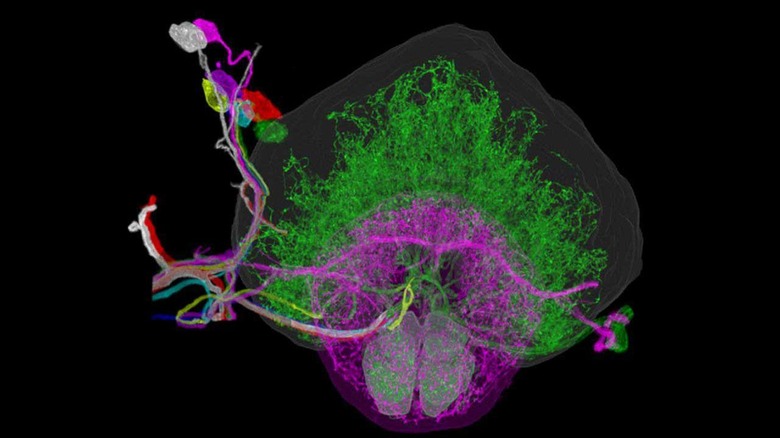
The discovery demonstrated that, under these constraints, light’s natural tendency toward disorder could reverse, producing an ultrafast beam without the need for additional shaping components. Researchers reported that the method generated 3D images of the blood-brain barrier at speeds about 25 times faster than two-photon microscopy while preserving similar image quality.
Real-Time Drug Tracking: A New Lens on Neurological Treatments
The blood-brain barrier—a selective membrane that protects the brain from harmful substances—has posed challenges in developing treatments for neurological diseases. Many potential therapies fail not because they lack effectiveness but because researchers cannot confirm whether the drugs reach their intended targets. The MIT team’s imaging technique could provide a way to observe drug interactions with the blood-brain barrier in real time.
The researchers explained that the method might help scientists determine whether treatments for conditions like Alzheimer’s or ALS successfully penetrate the brain. Beyond diagnostics, the ability to visualize drug interactions at a cellular level could inform adjustments to delivery mechanisms, dosages, or treatment strategies, all without the trade-offs in speed or resolution that have limited existing techniques.
A comparison with traditional methods highlights the advancement. Two-photon microscopy, while detailed, is relatively slow, often requiring minutes to capture a single high-resolution image. The new approach achieves comparable clarity in seconds, making it possible to track dynamic processes like drug absorption as they occur. This shift from static snapshots to continuous observation could reveal patterns that were previously difficult to detect.
The Engineering Trade-Offs: Speed, Precision, and the Limits of Control
The discovery emerged from an unexpected observation. While testing the limits of a fiber shaper, researchers noticed the laser’s behavior defied conventional expectations. Typically, increasing power causes light to scatter more due to imperfections in the fiber. Instead, as the power approached the fiber’s damage threshold, the light concentrated into a single, sharp beam.
This finding underscores a key tension in optical engineering: balancing control with adaptability. Multimode fibers, which can carry high-power lasers, are prone to scattering because of microscopic imperfections. Overcoming this disorder usually requires complex beam-shaping components, which add cost and complexity. The MIT team’s approach bypasses these challenges by leveraging the fiber’s inherent instability. At high power, the light reorganizes into a stable, ultrafast beam.
However, the method has limitations. The requirement for a zero-degree entry angle demands precise alignment, which could complicate use in clinical settings. Additionally, the reliance on high-power lasers raises questions about heat generation and long-term tissue safety. The researchers acknowledged these trade-offs but suggested they could be addressed through further engineering. They noted that while disorder is intrinsic to these fibers, the self-organizing effect could eliminate the need for custom beam-shaping components.
What to Watch: Barriers, Uncertainty, and the Path Forward
The immediate applications of this discovery are likely to be in research settings rather than clinical environments. The imaging method’s ability to track drug absorption in real time could expedite preclinical studies, offering a faster way to evaluate potential treatments for neurological diseases. However, translating this breakthrough into a widely used clinical tool will require overcoming several challenges, including regulatory approvals and integration with existing medical systems.

One of the primary questions is whether the technique can be adapted for real-world use. The zero-degree alignment requirement, while manageable in a lab, could prove difficult in settings where equipment is subject to movement or wear. Additionally, the long-term effects of high-power laser exposure on living tissue remain uncertain. While the method has been tested on the blood-brain barrier, its safety and effectiveness for other imaging applications, such as deep-brain scans, have not yet been established.
For now, the MIT team is focused on refining the technique and exploring its potential. Collaborators from other institutions are already testing the method in drug development studies, with early results expected to provide insights into its practical utility. If successful, the approach could transform how researchers study the brain, offering a tool that is not only faster but also fundamentally different from existing methods.
The discovery challenges a long-standing assumption in optical physics: that disorder and precision cannot coexist. By pushing the limits of high-power lasers, the researchers revealed an unexpected behavior of light. The next step is determining how far this principle can be applied and what new possibilities it may unlock.