Future Trends in HPV Research: Plasmids, Bulk Antigens, and Pentamers
The Evolving Role of Plasmids in HPV Research
Plasmids have long been a cornerstone in the development of HPV vaccines. The use of HPV L1/L2-expressing plasmids, such as p16LLw18p18llw19p6llw220 and p11Lw21, has been instrumental in advancing our understanding and treatment of HPV. These plasmids, provided by the National Cancer Institute, are crucial for expressing viral proteins essential for vaccine development. As research progresses, the focus on optimizing plasmid designs and expression systems will continue to drive innovation in HPV vaccine technology.
Advances in HPV Bulk Antigen Production
The production of HPV bulk antigens is a critical step in vaccine development. Companies like Beijing Health Guard Biotechnology and Innovax Biotech Co., Ltd. have been at the forefront of producing monovalent antigen bulks without aluminum adjuvant. These antigens, produced in various expression systems such as E. coli, yeast, and insect cells, are pivotal for developing effective vaccines. The future will likely see a shift towards more efficient and scalable production methods, ensuring a steady supply of high-quality antigens for vaccine manufacturing.
The Significance of HPV Final Products and Pentamers
HPV final products, such as VLPs (Virus-Like Particles) adsorbed on aluminum adjuvant, are the culmination of extensive research and development. Companies like Ruike Biotechnology and Bowei Biotechnology have been instrumental in providing these final products. The use of VLPs in vaccines mimics the structure of the virus without its harmful components, making them safe and effective. Pentamers, derived from these VLPs, are essential for understanding the structural and functional aspects of HPV proteins, paving the way for more targeted and effective treatments.
Innovations in Single Memory B Cell Isolation and Sequencing
The isolation and sequencing of single memory B cells from vaccinated individuals offer profound insights into the immune response to HPV vaccines. This process, involving the use of CD19 MicroBeads and flow sorting techniques, allows for the identification of specific B cells that produce neutralizing antibodies. By sequencing these cells, researchers can clone and study the antibodies, leading to the development of more effective vaccines and therapeutic agents.
Antibody Expression and Fab Generation
The rapid amplification of cDNA ends (RACE) technique has revolutionized the way antibodies are expressed and generated. This method allows for the synthesis of cDNA from mRNA, followed by the amplification of variable regions of the antibody. The resulting monoclonal antibodies can be processed to obtain Fab fragments, which are smaller and more versatile for various applications. This technology is crucial for developing targeted therapies and diagnostics for HPV.
Determining Antibody Binding Activities
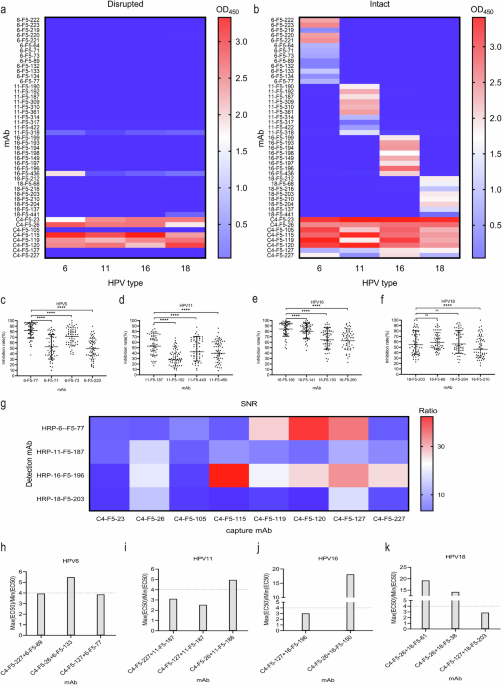
The determination of antibody binding activities is essential for evaluating the efficacy of HPV vaccines. Techniques such as ELISA (Enzyme-Linked Immunosorbent Assay) and screening ELISA for conformation-dependent binding provide valuable data on how well antibodies bind to HPV antigens. These methods are crucial for ensuring that vaccines elicit a strong immune response, protecting individuals from HPV infection.
Detection of Pseudovirus Neutralization
Pseudovirus neutralization assays are vital for assessing the neutralizing activities of monoclonal antibodies and serum samples. By using pseudoviruses, researchers can safely and effectively measure the ability of antibodies to inhibit viral infection. This method is crucial for developing and testing new vaccines and therapeutic agents against HPV.
Antibody Competition Assays
Antibody competition assays, such as those based on the Luminex platform, provide insights into the competitive binding of antibodies to HPV antigens. These assays help identify dominant neutralizing epitopes, which are critical for vaccine design. By understanding how different antibodies compete for binding sites, researchers can develop more effective vaccines that target multiple epitopes.
Validation of IVRP Methodology
The validation of IVRP (In Vitro Potency Assay) methodology ensures the reliability and accuracy of potency measurements for HPV vaccines. This process involves diluting and testing reference and test solutions to determine relative titers. The validation of IVRP methods is essential for maintaining the quality and efficacy of HPV vaccines.
Cryo-EM and Structural Analysis
Cryo-electron microscopy (Cryo-EM) and structural analysis techniques, such as homology modeling and real-space refinement, provide detailed insights into the structure of HPV proteins and their interactions with antibodies. These methods are crucial for understanding the molecular basis of HPV infection and for developing targeted therapies.
In Vivo and In Vitro Potency Assays
In vivo and in vitro potency assays are essential for evaluating the effectiveness of HPV vaccines. These assays involve testing the immune response in animal models and in cell cultures, respectively. The results from these assays provide valuable data on the potency and efficacy of HPV vaccines, ensuring their safety and effectiveness.
Future Directions in HPV Research
The future of HPV research is promising, with ongoing advancements in plasmid technology, antigen production, and structural analysis. As researchers continue to unravel the complexities of HPV, we can expect to see more effective vaccines and therapeutic agents that protect against this widespread infection.
FAQ Section
Q: What are HPV L1/L2-expressing plasmids, and why are they important?
A: HPV L1/L2-expressing plasmids are genetic constructs that express viral proteins essential for vaccine development. They are crucial for producing antigens that mimic the structure of the HPV virus without its harmful components.
Q: How are HPV bulk antigens produced?
A: HPV bulk antigens are produced using various expression systems, including E. coli, yeast, and insect cells. These antigens are then used to develop vaccines and other therapeutic agents.
Q: What are HPV final products, and why are they significant?
A: HPV final products, such as VLPs adsorbed on aluminum adjuvant, are the culmination of extensive research and development. They are significant because they mimic the structure of the virus, making them safe and effective for vaccination.
Q: How are single memory B cells isolated and sequenced?
A: Single memory B cells are isolated using CD19 MicroBeads and flow sorting techniques. They are then sequenced to identify specific antibodies that produce neutralizing responses, which are crucial for vaccine development.
Q: What is the RACE technique, and how is it used in antibody expression?
A: The RACE technique is used to synthesize cDNA from mRNA, followed by the amplification of variable regions of the antibody. This method is crucial for expressing and generating monoclonal antibodies and their Fab fragments.
Q: How are antibody binding activities determined?
A: Antibody binding activities are determined using techniques such as ELISA and screening ELISA for conformation-dependent binding. These methods provide valuable data on how well antibodies bind to HPV antigens.
Q: What are pseudovirus neutralization assays?
A: Pseudovirus neutralization assays are used to measure the ability of antibodies to inhibit viral infection. These assays are crucial for developing and testing new vaccines and therapeutic agents against HPV.
Q: What are antibody competition assays, and why are they important?
A: Antibody competition assays help identify dominant neutralizing epitopes, which are critical for vaccine design. These assays provide insights into how different antibodies compete for binding sites on HPV antigens.
Q: How is IVRP methodology validated?
A: IVRP methodology is validated by diluting and testing reference and test solutions to determine relative titers. This process ensures the reliability and accuracy of potency measurements for HPV vaccines.
Q: What is Cryo-EM, and how is it used in HPV research?
A: Cryo-EM is a technique used to study the structure of HPV proteins and their interactions with antibodies. It provides detailed insights into the molecular basis of HPV infection and helps in developing targeted therapies.
Q: What are in vivo and in vitro potency assays?
A: In vivo and in vitro potency assays are used to evaluate the effectiveness of HPV vaccines. These assays involve testing the immune response in animal models and in cell cultures, respectively, providing valuable data on the potency and efficacy of vaccines.
Did You Know?
HPV vaccines have been instrumental in reducing the incidence of cervical cancer, a disease primarily caused by HPV infection. The development of effective vaccines has saved countless lives and continues to be a critical area of research.
Pro Tips
Tip 1: Stay informed about the latest advancements in HPV research to ensure you are up-to-date with the most effective prevention and treatment strategies.
Tip 2: Regular screenings and vaccinations are key to preventing HPV-related diseases. Encourage friends and family to get vaccinated and undergo regular screenings.
Tip 3: Support ongoing research and clinical trials to contribute to the development of more effective HPV vaccines and therapeutic agents.
Call to Action
We invite you to share your thoughts and experiences with HPV vaccines in the comments section below. Your insights can help others understand the importance of vaccination and encourage further research. For more information on HPV and related topics, explore our other articles or subscribe to our newsletter to stay updated on the latest developments in medical research.