For the first time, researchers have succeeded in correcting defective genes directly in human skin, using a locally applied therapy. The discovery paves the way for completely new treatments for genetic skin diseases, for which there are currently no curative solutions.
A team of researchers from the University of British Columbia (UBC), Canada, in collaboration with the Berlin Institute of Health at Charité, from Germany, has developed the first gene therapy capable of correcting mutations in human skin by direct application.
The results were published on Tuesday in the magazine Cell Stem Cell and describe an approach that targets the genetic cause of the disease, not just the symptoms.
In the study, the researchers tested the therapy on models obtained from living human skin. The treatment was designed to correct the most common genetic mutation responsible for autosomal recessive congenital ichthyosis (ARCI), a rare and severe disease present from birth.
The condition occurs in approximately one in 100,000 people and is characterized by extremely dry and scaly skin, chronic inflammation and increased risk of infection.
Currently, there is no effective treatment to alter the course of the disease, and patients are forced to manage their symptoms throughout their lives.
The application of the therapy allowed the recovery of up to 30% of the normal function of the skin, a threshold considered clinically relevant based on the results reported in previous studies.
According to the authors, even a partial correction of the genetic defect can produce a significant improvement in skin function.
Although congenital ichthyosis affects a small number of patients, the researchers point out that the strategy used can be adapted for other genetic skin diseases.
These include epidermolysis bullosa, a serious condition characterized by extreme skin fragility, as well as more common diseases such as eczema or psoriasis.
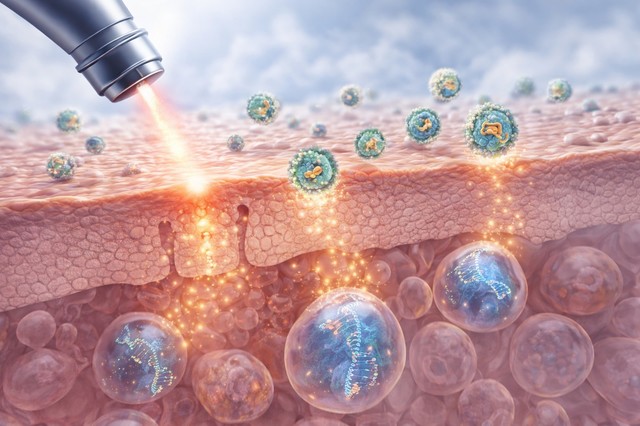
A key element of the research is the method of delivery of gene editing technology.
The skin acts as a protective barrier, which makes it difficult for large biological therapies to penetrate. To overcome this obstacle, the team used lipid nanoparticles, microscopic structures that can carry genetic material inside cells. This technology is already known for its use in messenger RNA (mRNA) vaccines.
Before applying the therapy, the researchers made painless micro-openings in the superficial layers of the skin using a clinically approved laser. Through these openings, the lipid nanoparticles reached the stem cells of the skin. Once inside the cells, the genetic editor corrected the mutation in the DNA, allowing the skin to begin functioning closer to normal.
According to the authors, the treatment has a strictly local effect on the skin, with no evidence of unwanted genetic changes in other areas of the body.
This aspect is considered essential for the safety of a possible clinical use.
The study was carried out in collaboration with Vancouver-based biotechnology company NanoVation Therapeutics, which specializes in the development of genetic drugs based on lipid nanoparticles.
The therapy is not yet approved for clinical use, but researchers are currently working with regulatory authorities to establish the necessary steps for clinical testing in humans.
The objective in mind is the initiation of the first clinical trials on patients, with the aim of developing a safe and effective treatment for people who currently have no real therapeutic options.