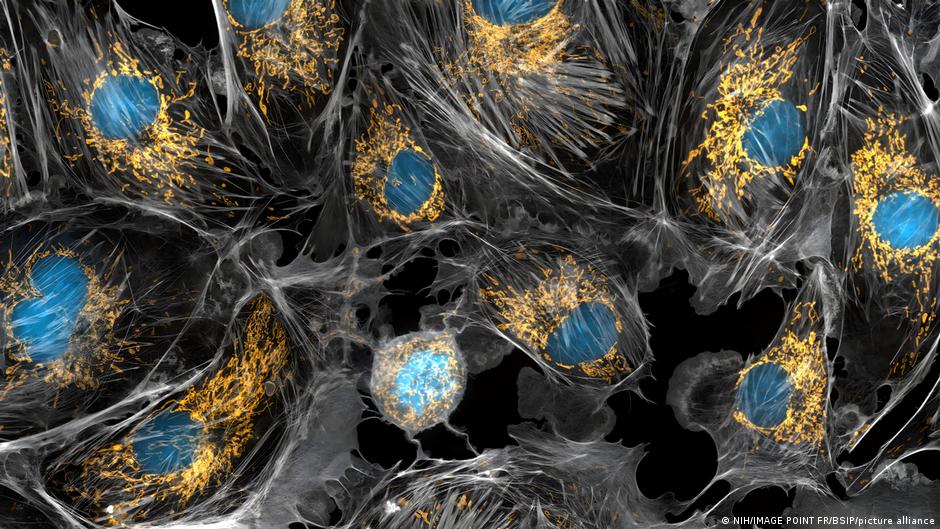
A team of biomedical researchers from Texas A&M University in the United States has developed an innovative method to restore vitality to damaged or aging human cells, a preliminary advance that could open new avenues in the treatment of diseases linked to cellular deterioration.
The key to the discovery is in the mitochondria, those tiny structures that function as the “batteries” of the cell. With the passage of time – and under attacks such as neurodegenerative disorders or certain oncological treatments – these energy plants lose number and efficiency. This loss has widespread repercussions: without sufficient internal fuel, cells see their functioning and repair capacity limited.
Nanoflowers and stem cells: a promising experimental approach
Table of Contents
Faced with this challenge, the team led by Dr. Akhilesh K. Gaharwar and PhD student John Soukar proposes an alternative approach that avoids genetic modifications or complex drug combinations. Their method is based on flower-shaped particles, known as nanoflowers, which are applied directly to stem cells.
Made with molybdenum disulfide, an inorganic compound that presents unique properties on a microscopic scale, these structures – as pointed out Science Alert– they work like real sponges capable of eliminating harmful oxygen molecules. At the same time, they activate genes that drive the production of new mitochondria within stem cells.
The effect is remarkable. As Texas A&M University points out, Treated stem cells are transformed into true “mitochondrial biofactories”, capable of generating up to twice as many mitochondria as untreated cells.
And there is even a more striking twist. When these “superfed” cells were placed next to damaged or aged cells, they transferred between two and four times more mitochondria, sharing part of their energy abundance with their needier neighbors.
“We have trained healthy cells to share their spare batteries with weaker ones,” Gaharwar explains in the press release. “By increasing the number of mitochondria within donor cells, we can help aged or damaged cells regain their vitality, without the need for genetic modifications or medications,” he adds.
“It’s like giving an old electronic device a new battery,” explains Soukar. “Instead of throwing them away, we are connecting fully charged batteries from healthy cells to sick ones.”
The team, which published its study in Proceedings of the National Academy of Sciences, tested the method on muscle and heart cells exposed to chemotherapy – agents notoriously harmful to cellular health – and observed that those treated with enhanced stem cells resisted damage better and maintained their energetic activity.
Possible medical applications
Possible applications – if the following steps confirm its safety – cover conditions in which mitochondrial failure is relevant, such as certain neurodegenerative disorders, cardiomyopathies, muscular dystrophies and genetic mitochondrial diseases.
“(The cells) could be placed anywhere on the patient,” Soukar says. “Thus, in the case of cardiomyopathy, cardiac cells can be treated directly, placing the stem cells directly in or near the heart.”
Anti-aging method?
Gaharwar, along with Soukar and an interdisciplinary team, clarifies that this advance should not be interpreted as an anti-aging panacea, although certain aspects of aging linked to mitochondrial decline could benefit.
They also point out that, in complex diseases such as Alzheimer’s, an improvement in mitochondrial health may be able to slow down degeneration, but it is still early to say that it will completely reverse these pathologies. All in all, this is a promising step in regenerative medicine.
Since it is based on a natural process – the transfer of mitochondria already occurs spontaneously in the body, although on a much smaller scale – this method does not introduce mechanisms outside the body, but rather amplifies those that already exist.
An additional advantage is that these nanoparticles remain inside cells longer than conventional drugs, which could reduce the frequency of administration and sustain mitochondrial biogenesis without constant interventions.
Next steps towards clinical trials in regenerative medicine
The researchers emphasize, however, that there is still a long way to go. As reported Newsweekthe next steps include evaluating the technique in animal models, analyzing its safety and verifying its long-term effectiveness before considering clinical trials in humans.
Still, the door opens to a new type of medicine in which the body’s own cells, equipped with microscopic tools, collaborate with each other to stay alive and functional.
Soukar sums it up with an optimistic phrase: “It’s just the beginning. We could work on this indefinitely and discover new things and new treatments for diseases every day.”
Edited by Felipe Espinosa Wang with information from Texas A&M University, PNAS, Science Alert and Newsweek.